Our human-derived ECM (hECM) hydrogel represents a breakthrough in creating physiologically relevant extracellular environments for advanced cell culture and disease modeling. Developed from native human tissues, hECM retains the essential biochemical cues and structural proteins necessary to closely replicate the natural human microenvironment.

We achieved this by carefully developing a proprietary decellularization and processing method that preserves the native composition and functional integrity of key ECM proteins, including collagen, elastin, laminin, and fibronectin. By avoiding harsh chemical treatments and optimizing extraction conditions, we maintained the bioactivity of essential signaling molecules and structural components critical for supporting cell adhesion, proliferation, and differentiation. Additionally, we fine-tuned the hydrogel’s mechanical properties to closely mimic physiological stiffness, enabling cells to sense and respond to their environment naturally.

Overcoming the limitations of traditional ECM reconstruction
Traditional ECM research often focuses on isolated protein analysis through proteomics and in vitro studies using generic scaffolds. At Preci, we aimed to unify these areas by creating a native human-derived ECM matrix. Previous efforts, using pepsin digestion, broke down key both structural and signaling proteins, limiting the ability to generate functional ECM systems.

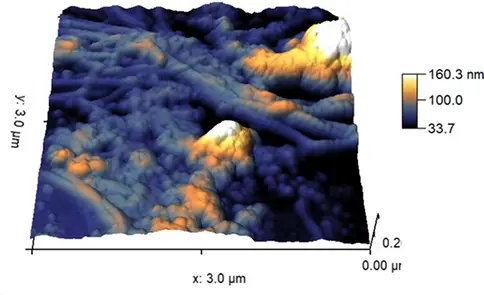
SOL
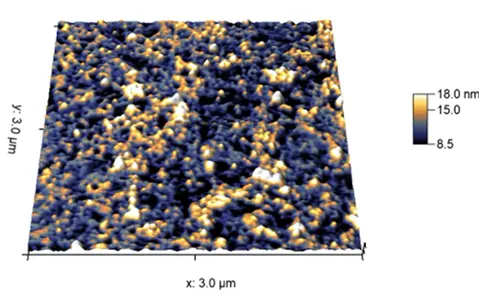
GEL

Native tissue ECM systems plays not only supporting role for the structural integrity of the tissue, it is also essential component of the signal transduction. Integrin-dependent pathways drive a lot of cellular physiology reactions via impacting signalling. Examples of such signalling routes include EGFR-pathway, MAPK/ERK-pathway, PI3K-pathway, and apoptotic pathways. Apart from that, many cells-matrix interactions frequently navigate tissue behaviour via the local contraction or release of the ECM proteins. Cell-matrix interactions are crucial in modelling of cellular adhesion, cell-cell interactions, cellular invasion, migration, vascularisation and much more.

Preci has redefined the state-of-the-art in ECM research with the introduction of our hECM isolation procedure, an innovative ECM dispersion created through the gentle swelling of ECM proteins. ExViGel represents a significant advancement, preserving the native structure and functionality of ECM components. This breakthrough product is now accessible to the research community, offering a reliable and scalable solution with multi-liter production capability and validated continuous production processes.

Our ECM hydrogel is uniquely designed to support self-assembly directly with cells, enabling the formation of complex, tissue-like structures without the need for external scaffolding. Cells expressing alpha-SMA (α-Smooth Muscle Actin)demonstrate particularly high potency in driving this self-organization, forming stable cellular sheets embedded within the hydrogel matrix. Upon TGF-beta stimulation, these sheets exhibit functional contractility, closely mimicking in vivo tissue remodeling and fibrosis-related processes. This makes our ECM hydrogel an ideal tool for advanced studies in tissue engineering, fibrosis modeling, and mechanobiology.


