The fluid nature of tissue was recognized through the groundbreaking work of Phillips and Steinberg in 1978 [1]. They confirmed the viscoelastic properties of cell aggregates during embryogenesis, uniting evidence for both the fluid and elastic solid hypotheses. Since then, we have learned much more about the spatiotemporal organization of tissues and the molecular mechanisms that underlie it.
However, several recent works by Amy E. Shyer and Alan R. Rodriguez [2-3] have provided much deeper insight into how molecular events shape macroscopic tissue development. Tissue organisation phenomena such as symmetry breaking and pattern formation (e.g., follicular budding) are driven by cell-matrix interactions and tuned by molecular cascades that increase or reduce fluidity. The term “supracellular fluidity” was coined in this series of works and seems to have an impact far beyond fundamental, curiosity-driven discoveries.
Symmetry breaking in living systems is a long-known phenomenon. Yakov Zeldovich first discovered reaction-diffusion instability in combustion reactions, and Alan Turing later applied it to explain pattern formation during morphogenesis. Reaction-diffusion behavior has also been observed in key morphogenesis drivers such as FGF, BMP, and Shh.
Palmquist et al [2] observed a similar type of symmetry breaking in a surprisingly simple cellular system. Chicken skin fibroblasts, when mixed with collagen and dispensed in a circular pattern into a well plate, rapidly (within 48 hours) reduce symmetry from radial to discrete polygonal patterns. The homogeneous cell population and the absence of an initially imposed hormone gradient supported the conclusion that instability in this system is caused by contractile interactions between cells and the extracellular matrix. Additionally, the authors showed that calcium signalling tunes the system’s behavior, affecting both the shape and the mechanical properties of the resulting buds.
Later, S. Yang et al. showed [3] that gradients of fibroblast growth factors (FGF) and bone morphogenetic protein (BMP) support budding in vivo. FGF acts as a “tightener”, while BMP acts as a “softener” of the cell-ECM complex. Specifically, BMP signalling activation increases the “fluidity” of the system by reducing the elastic modulus and increasing the viscosity of the supracellular system. Interestingly, contractility also increases with BMP pathway activation.
Of course, these findings shed light on the fundamental nature of tissue mechanics, but they also raise many questions, especially regarding relevance to human disease. For example, BMP is a well-known growth factor in the development of many tumors, driving cell migration and invasion [4]. However, this complex behavior is shaped by many pathways acting simultaneously (e.g. TGF-beta family, Wnt/b-catenin pathway). The essential question, then, is whether investigating symmetry breaking and “fluidity” can be used to model the invasive potential of tumors.

Figure 1. Mechano-biological descriptors used by Amy E. Shyer and Alan R. Rodriguez to study the interplay between morphogen effects and the spatiotemporal organisation of tissue
Mechano-biological tissue properties are also highly relevant to fibrosis and chronic disease [5]. Tissue stiffening and the resulting loss of function, driven by fibroblast accumulation and ECM deposition, are central to this pathophysiological process. Therefore, tissue contractility and cellular motility can serve as in vitro biomarkers of fibrosis development. TGF-b signalling is a key driver of this process. Although it often acts through the same SMAD transduction pathways as BMP, TGF-b signalling promotes ECM protein accumulation, thereby stiffening the cell-ECM assembly.
In this context, we can pose a more blunt question:
Can we use measurable mechanobiological descriptors, such as viscosity, elastic modulus, and contractility, as in vitro biomarkers of disease progression?
In fact, to answer that question, we would need to recreate in vitro the interaction between ECM and contractile cells, as it occurs in the diseased state. Since real-world biological systems involve far more complex pathways and interactions, no simple system can simulate tissue “fluidity” effects in vitro. Primary cells and primary ECM are prerequisites for interrogating the connection between molecular network dynamics in disease and macroscopic changes in tissue.
Preci’s Human-Derived Extracellular Matrix models allow us to recreate native tissue contractility through the simple mixing of primary stromal cells (fibroblasts) and hECM. Due to strong adhesive interactions between cells and the ECM, homogeneous discs self-assemble. Earlier, we showed a straightforward approach to measuring the elastic modulus of these discs.
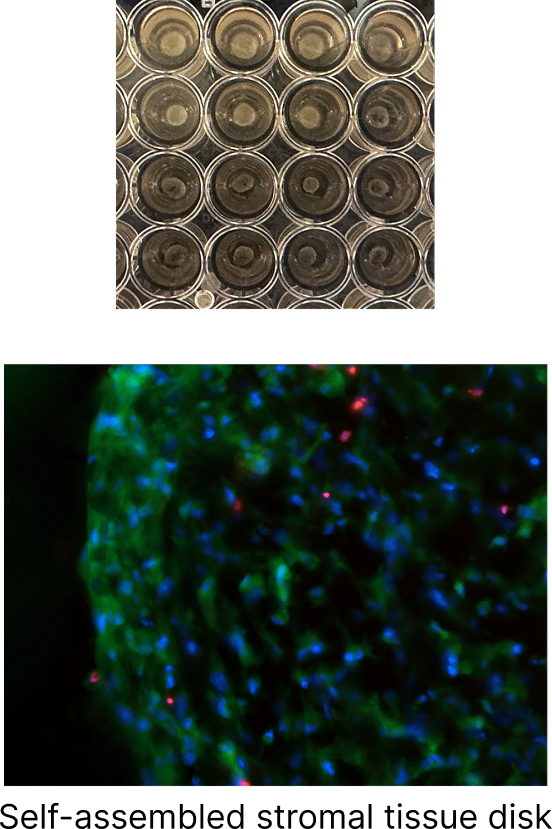
Figure 2. Self-assembly of the fibroblast–ECM complex drives contractile disc formation. Macroscopic and microscopic (DAPI/Calcein-AM/PI) images are shown.
These systems match the complexity of in vivo tissue assemblies by integrating the full, unaltered ECM proteome with cells extracted from the same donors. They therefore offer a complete system to answer the question raised above. Moreover, these systems can be derived from patient cohorts with pathophysiological conditions and from healthy controls to study the drivers and hallmarks of mechanobiological changes in the organ or tumor.
- Phillips HM, Steinberg MS. Embryonic tissues as elasticoviscous liquids. I. Rapid and slow shape changes in centrifuged cell aggregates. J Cell Sci. 1978 Apr;30:1-20. doi: 10.1242/jcs.30.1.1.
- Palmquist KH, Tiemann SF, Ezzeddine FL, Yang S, Pfeifer CR, Erzberger A, Rodrigues AR, Shyer AE. Reciprocal cell-ECM dynamics generate supracellular fluidity underlying spontaneous follicle patterning. Cell. 2022 May 26;185(11):1960-1973.e11. doi: 10.1016/j.cell.2022.04.023.
- Sichen Yang et al. ,Morphogens enable interacting supracellular phases that generate organ architecture.Science382,eadg5579(2023).DOI:10.1126/science.adg5579
- Ehata S, Miyazono K. Bone Morphogenetic Protein Signaling in Cancer; Some Topics in the Recent 10 Years. Front Cell Dev Biol. 2022 May 25;10:883523. doi: 10.3389/fcell.2022.883523.
- Ingo Ganzleben, Benjamin D. Medoff. Mechanobiology and the extracellular matrix in pulmonary fibrosis. iScience, 2025, https://doi.org/10.1016/j.isci.2025.113993.


