The Gel Contraction Assay (GCA) stands as a commonplace *in vitro* technique employed to gauge the capacity of cells to contract when situated within a three-dimensional (3D) extracellular matrix (ECM). Originally introduced by Bell in 1979 for the examination of how fibroblasts engage with their matrix, this procedure has since turned into an indispensable tool across mechanobiology, tissue regeneration, and investigations into matters like wound repair and scarring. The fundamental concept behind the assay involves placing cells within a supportive hydrogel framework—usually constructed from collagen or fibrin—and subsequently tracking the slow, visible shrinking of the gel brought about by the pulling forces exerted by these cells.
The actual contraction process relies upon the aptitude of cells exhibiting mechanical activity, chiefly fibroblasts and myofibroblasts, to anchor themselves onto matrix strands via focal attachments mediated by integrins, and subsequently exert forces through their internal structure, mainly driven by actomyosin tension. These generated forces propagate across the entire gel meshwork owing to its strong internal connections, resulting in a measurable decrease in the overall surface area or volume of the sample. The degree to which the gel shrinks serves as a straightforward, reagent-free measure of how actively the cells are contracting, their health, and how effectively they are interacting with the ECM; this can be easily quantified using standard bright-field visual inspection.
Materials and Technical Setup:
Reagents and consumables
• Extracellular matrix (ECM) hydrogel
• Cell culture medium (ice-cold, for ECM dilution)
• Fibroblast cell suspension
• Calcein AM
• Phosphate-buffered saline (PBS)
Equipment
• Water bath (≤37 °C)
• Ice bath
• Ultrasonic bath or vortex
• Micropipettes with ice-cold tips
• 96-well plate
• Centrifuge
• CO₂ incubator (37 °C, 5% CO₂)
• Cell counter
3D thick gel with cells in the ECM layer was achieved as described below:
1. Prepare the cells to be used in the experiment using the standard Thawing Cell protocol. Dilute them to a minimum volume.
2. Thaw the hydrogel vial rapidly until only a small crystal of ice remains using a water bath with a temperature of 37°C or less, avoiding secondary ice formation. Thawing should not take longer than 3-5 minutes before the hydrogel is homogenized.
3. While the hydrogel is still warm from thawing, mix it intensively by pipetting 20-30 times with an ice-cold tip, avoiding foaming. For better results, use an ultrasonic bath or vortex immediately after thawing.
4. Resuspend the gel in ice-cold culture medium. In this system, we recommend a dilution of 1/5 to 1/20. For a more homogenized solution, use an ultrasound or a vortex.
5. Add your cells to the diluted gel. Resuspend gently.
6. Add the required volume to the plate. For a 96-well plate, we recommend 200 μL of diluted gel per well.
7. Centrifuge at 4°C, 200 g, 10 minutes.
8. Incubate the plate at 37°C overnight, avoiding any agitation to prevent disturbing the gel.
ECM was diluted with cell culture medium to a minimal concentration 0,7 mg/mL and a maximal concentration of 2,8 mg/mL.
Upon the addition of fibroblasts, gels start to contract upon standing to various degrees.

The photo on the left is the first day after passaging the cells, and the photo on the right is the
third day after incubation.
Upon imaging of fibroblast layers cells were observed using calcein AM staining to visualize living cells.
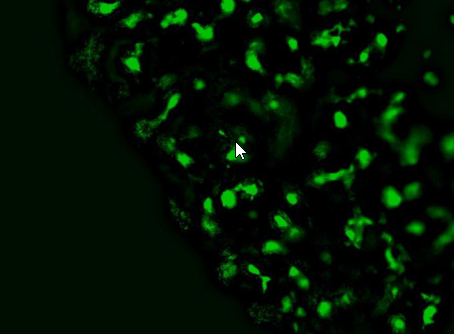
Imaging of disks with calcein on the 3rd day of incubation.
Contraction reached a minimum volume after 3 days of incubation. The contraction area decreases upon an increase in concentration and an increase in cell number. The following results were observed.
| Gel Concentration/Celnumber | 40,000 | 30,000 | 20,000 | 10,000 |
| 2,8 mg/mL | Contracted disc 95% of the well area | Contracted disc 100% of the well area | Contracted disc 100% of the well area | Contracted disc 100% of the well area |
| 1,4 mg/mL | Contracted disc 45% of the well area | Contracted disc 85% of the well area | Contracted disc 90% of the well area | Contracted disc 95% of the well area |
| 0,93 mg/mL | Contracted disc 25% of the well area | Contracted disc 30% of the well area | Contracted disc 50% of the well area | Contracted disc 90% of the well area |
| 0,7 mg/mL | Contracted aggregate, 2000 um in diameter | Contracted aggregate, 3000 um in diameter | Contracted disc 30% of the well area | Contracted disc 90% of the well area |
Frequently Asked Questions (FAQ)
1. Why does a loose aggregate form instead of a cohesive disc at ECM concentrations ≤0.7 mg/mL?
At low ECM concentrations (≤0.7 mg/mL), the fiber network density of the hydrogel is insufficient to maintain a cohesive gel structure against cellular traction forces. Instead of uniform compaction of the entire disc, cells pull the matrix into focal points, forming compact aggregates of 2000–3000 μmin diameter. To obtain a stable disc, it is recommended to increase the concentration to 0.93 mg/mL or higher, or to increase the cell seeding density to ≥20,000 cells per well.
2. What mechanical parameters can be measured in contracted gel discs?
A contracted disc represents a densified tissue-like construct whose mechanical properties differ substantially from those of the original hydrogel. Key measurable parameters include Young’s modulus (matrix stiffness), viscoelastic characteristics, and resistance to deformation. In particular, changes in the elastic modulus of the construct during and after contraction can be quantified using the micropipette aspiration technique described previously in the laboratory blog. In this method, an amicropipette tip with a calibrated inner diameter is brought into contact with the surface of the disc, and a controlled negative pressure is applied; the depth of material aspirated into the pipette in response to a known pressure allows the Young’s modulus of the construct to be calculated. Comparing modulus values across discs of varying ECM concentration and cell seeding density makes it possible to directly track how cell-driven matrix remodeling alters construct stiffness relative to a cell-free control.
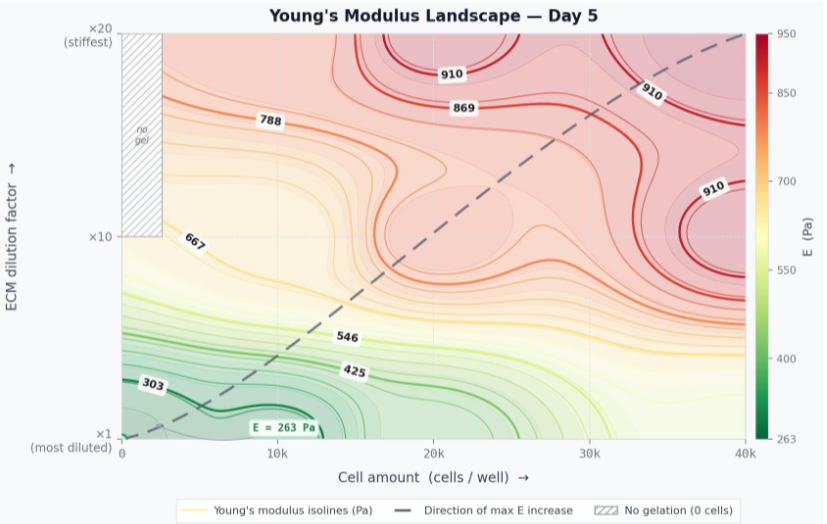
3. How do you understand the residual area percentage, and what is the way to compare different conditions?
The amount of contraction is shown as a percentage of the well area taken up by the gel on day 3 of the incubation compared to the starting area (100% means the whole well is covered). A lower value corresponds to a stronger contraction. When comparing different conditions, you need to consider both the ECM concentration and the number of cells at the same time. For example, when the concentration is 1.4 mg/mL, and there are 40,000 cells, the remaining area is about 45%, but at the same concentration with 10,000 cells, it’s around 95%. This shows that the number of cells planted plays a big role when the matrix concentration is moderate.
4. Is centrifugation required, and what occurs if it is not done?
Centrifuging at 4°C, 200 g for 10 minutes helps spread the cells evenly throughout the gel and ensures the matrix is in close contact with the bottom of the well before it hardens. If you don’t use centrifugation, the cells might settle in an uneven way, and the gel disc could form with some flaws in its structure. This can make the results less reliable and the construct less stable. Discs that are not spun in a centrifuge are easier to move around when changing the liquid, so it’s important to use a careful method when taking out the liquid.
5. How can you change the medium without damaging the disc?
Discs that are formed through contraction but not fixed with extra support are weak in terms of strength and can be moved around easily by the flow of liquid. When taking up the used medium, the pipette tip needs to be kept carefully against the side of the well, making sure it doesn’t touch the disc directly. Fresh medium should be added gradually, making sure the flow goes towards the wall instead of hitting the top of the structure. If the construct is very unstable — like when the ECM concentration is close to the lowest working level — it’s better to use a multichannel pipette with the lowest possible flow rate or to balance the pressure in the tip before releasing the liquid.


