Immunofluorescence may be challenging for 3D structures, as it differs from 2D adherent cells, which do not suffer from diminished diffusion through the multicellular aggregates. The most frequently encountered problem is that the fluorochrome-conjugated antibody does not distribute evenly within spheroids, remaining on the surface, so the core remains unstained.
Most of the time, antibody penetration into the center of the spheroid depends on the permeabilization technique, which provides a pathway through the membrane holes. On the one hand, the staining duration should allow the diffusion of the antibodies throughout the confined cellular and ECM mass. On the other hand, the staining should not take too long, so that cells do not start to lose their morphology and decompose.
This protocol offers tips and advice for better spheroid IF.
Materials and Equipment:
• 0.22 um filters
• Syringes
• Micropipettes and tips
• Fridge
• Foil
• Laminar Box
• 50ml test tubes
• Microtubes
• Multimode plate reader
Reagents:
• 1X Phosphate Buffered Saline (PBS)
• 4% Formaldehyde in 1X PBS
• 0.5% Triton X-100 in 1X PBS
• 1% BSA in 1X PBS
• Antibody
• DAPI Fluorescence Mounting Medium
Steps of the process:
Note: Filter all the solutions made in PBS with a syringe through a 0.22 µl filter to avoid mechanical inclusions and microorganisms, which can cause background signal in wells.
Fixation
1. Aspirate culture medium from wells and rinse spheroids with PBS twice.
2. Fix spheroids with 4% formaldehyde for 25 min at room temperature.
3. Wash the wells with PBS three times for 5 min each.
Permeabilization
4. Add to wells 0.5% Triton X-100 and incubate for 20 min at room temperature.
5. Wash the wells with PBS thrice for 5 min each.
Blocking
6. Incubate spheroids with 1% BSA for 40 min at room temperature.
Staining
7. Aspirate blocking buffer and incubate spheroids overnight in a needed dilution of an antibody in 1% BSA at 4°C. It is important to cover your plate with foil so that light will not ruin the fluorochrome.
8. Aspirate staining buffer and wash wells with PBS thrice for 5 min each.
9. Aspirate all the PBS and stain the nucleus with DAPI Fluorescence Mounting Medium (with only one drop of medium on the well) for 5 min at room temperature in a dark place.
10. Read the plate with the multimode plate reader.
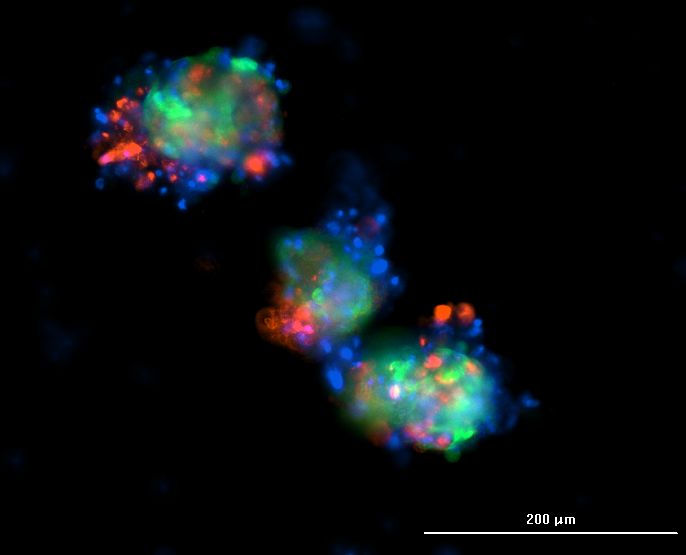
Spheroid protocols are constantly being refined as new technologies emerge. To stay updated on the latest breakthroughs in 3D cell culture, imaging optimization, and automated workflows, explore our Insights & Innovations blog.


