Simple reliable assays render drug that are clinically useful.
We all recall the successful attempt of James Cook to discover the drug against scurvy. He basically conducted the first placebo-controlled clinical study known to humanity.
Discovery of synthetic and semi-synthetic antibiotics wouldn’t be possible without the predictive nature vitro bacteria cultivation. In this way human being realized the potential of drug testing without jumping straight into clinical trials.
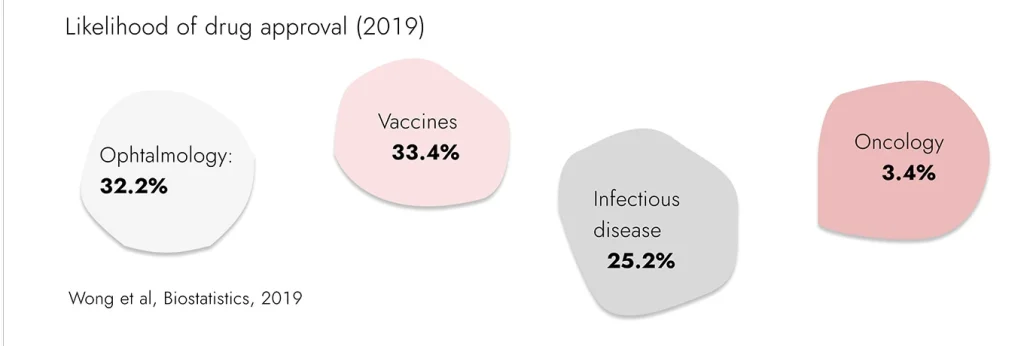
Unfortunately, this approach cannot be applied to all disease areas. Oncological drugs for many years already has the worst success rate in clinical studies. Translation from conventional cell and animal models of cancer diseases is still at the lowest level in drug discovery.
Conventional immortalized cell lines, despite being highly scalable, fail to represent the human disease. Perhaps the genomic instability of those during prolonged cultivation alters the response of the cell to the applied drug. Another problem is that real human tissue has much higher complexity than monoclonal cell culture.
Modern organoid, spheroid, and organ-on-chip cultures attempt to resolve those issues.
Preci is a platform that wants to unite those systems and use only the most relevant ones (hystologically and genetically) to bring more representative drug discovery. Additionally, it has the capacity to derive those cell models from the population of donors (10s and 100s for some diseases).
Every single patient needs to be represented in vitro to accomplish unbiased conclusions. No technical solution offers a generalized approach to model all cancer cases. Our case-to-case approach and thorough predictive analysis, involving real patient history, can achieve that.
Discover how Preci’s human-derived cell models deliver precise, patient-matched predictions for cancer therapies. Contact our team today for donor model access or predictivity consultation.


