ADME (Absorption, Distribution, Metabolism and Excretion) are the processes that determine how drugs move through the body. DMPK (Drug Metabolism and Pharmacokinetics) combines these processes with toxicity studies to figure out if a drug is safe, how much of it to give, and if it will work. Poor ADME properties cause about 40% of clinical failures.
ADME strategies continue to evolve alongside the emergence of new therapeutic modalities. At Preci, our efforts in the ADMET field focus on systematizing available in vitro approaches and delivering a comprehensive toolbox for permeability, distribution, metabolism, excretion, and toxicity studies. We stay aligned with the ongoing progress in the field, offering a broad product portfolio that spans from classical assay systems, such as human hepatocytes and microsomes, to models tailored for novel chemical entities. In particular, we dedicate special attention to developing ADME solutions for peptides and biologics. Most of the assay systems we offer are based on human-derived materials. Through years of experience, we have developed rigorous and ethical approaches to working with human tissues. Finally, our quality assurance process directly aligns product release testing with commonly used assay validation workflows. This approach minimizes discrepancies between scientists’ expectations and our quality control data.
We are always open to running a new assay or reference compounds set, validating our product for the specific demand. This brochure summarizes all technical data about our products and surveys our capabilities devoted to in vitro ADMET scientists.
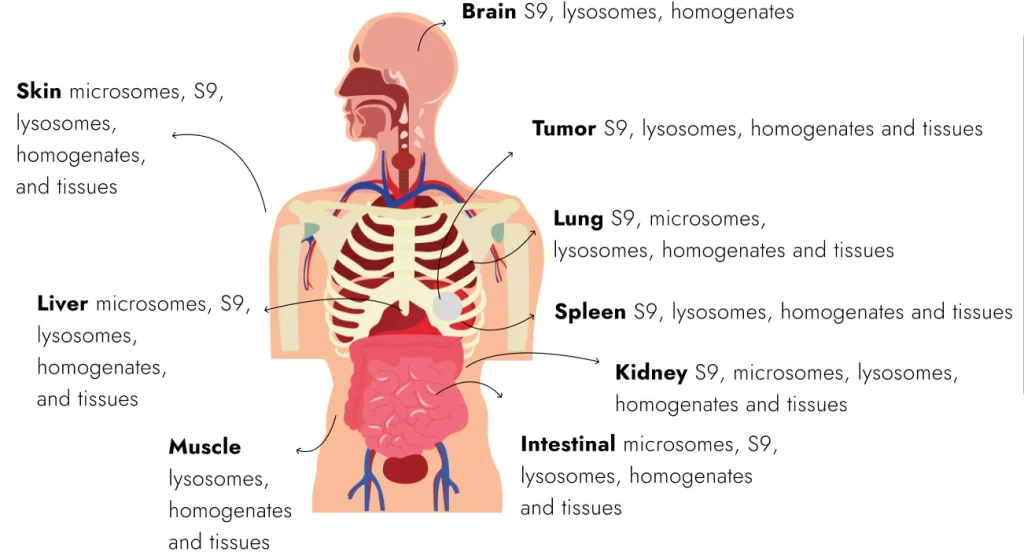
Our ADME and DMPK products portfolio keeps growing, and we are excited to share the latest updates in our freshly redesigned brochure. Inside, you will find an expanded product portfolio with new cell and subcellular models, along with clear data summaries that make it easier to find what you need for your DMPK research. This brochure reviews the products by their intended application, for easier navigation, and intends a focused review.


